Atom Vs Molecule Size
Atom Vs Molecule Size. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. A molecule consists of two or more atoms that can be either of the same or different elements.
Nejlepší Molecule Wikipedia
An atom is the smallest unit of matter. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. The covalent radii of the main group elements are given in the figure below. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures).Definitions of the atomic radius.
Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. A molecule consists of two or more atoms that can be either of the same or different elements. Molecules, on the other hand, can be …

Definitions of the atomic radius... While atoms are spherical in nature, molecules may be triangular, linear, or angular. (b) the metallic atomic radius, rmet, is. It is an indication of the size of the molecule as a target. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. These data confirm the trends observed for metallic …

1 å = 1 × 10−10 m = 100 pm... These radii are generally not the same (part (d) in figure 2.8.2). 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. These data confirm the trends observed for metallic … 27.05.2019 · atomic number atomic radius in nanometers; While atoms are spherical in nature, molecules may be triangular, linear, or angular. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Shape is another notable difference between the two. A molecule consists of two or more atoms that can be either of the same or different elements.. Definitions of the atomic radius.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound... An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. 1 å = 1 × 10−10 m = 100 pm. 27.05.2019 · atomic number atomic radius in nanometers; These radii are generally not the same (part (d) in figure 2.8.2). It is an indication of the size of the molecule as a target. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. Definitions of the atomic radius. An atom is the smallest unit of matter. Shape is another notable difference between the two.
An atom is the smallest unit of matter... The covalent radii of the main group elements are given in the figure below... A molecule consists of two or more atoms that can be either of the same or different elements.

These data confirm the trends observed for metallic … Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. Definitions of the atomic radius. An atom is the smallest unit of matter. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged.. 1 å = 1 × 10−10 m = 100 pm.
25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. An atom is the smallest unit of matter. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. 1 å = 1 × 10−10 m = 100 pm.

Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule... A molecule consists of two or more atoms that can be either of the same or different elements.

Shape is another notable difference between the two... (b) the metallic atomic radius, rmet, is. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. The covalent radii of the main group elements are given in the figure below. It is an indication of the size of the molecule as a target. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus.

These data confirm the trends observed for metallic … These radii are generally not the same (part (d) in figure 2.8.2). 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. These data confirm the trends observed for metallic … A molecule consists of two or more atoms that can be either of the same or different elements. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size.. It is an indication of the size of the molecule as a target.

An atom is the smallest unit of matter. These data confirm the trends observed for metallic … 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2... 27.05.2019 · atomic number atomic radius in nanometers;

An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus... 27.05.2019 · atomic number atomic radius in nanometers; The covalent radii of the main group elements are given in the figure below. It is an indication of the size of the molecule as a target. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. Definitions of the atomic radius. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule.. Definitions of the atomic radius.

An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Molecules, on the other hand, can be … 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. (b) the metallic atomic radius, rmet, is. The covalent radii of the main group elements are given in the figure below. An atom is the smallest unit of matter. Shape is another notable difference between the two. These data confirm the trends observed for metallic … (b) the metallic atomic radius, rmet, is.

Molecules, on the other hand, can be … Molecules, on the other hand, can be … A molecule consists of two or more atoms that can be either of the same or different elements. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. 1 å = 1 × 10−10 m = 100 pm. These data confirm the trends observed for metallic ….. The covalent radii of the main group elements are given in the figure below.
(a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. A molecule consists of two or more atoms that can be either of the same or different elements. 1 å = 1 × 10−10 m = 100 pm. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. 27.05.2019 · atomic number atomic radius in nanometers;. 1 å = 1 × 10−10 m = 100 pm.

(a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. It is an indication of the size of the molecule as a target. Definitions of the atomic radius. Shape is another notable difference between the two. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. 1 å = 1 × 10−10 m = 100 pm. Molecules, on the other hand, can be … These radii are generally not the same (part (d) in figure 2.8.2). The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. An atom is the smallest unit of matter.. (b) the metallic atomic radius, rmet, is.

(a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. (b) the metallic atomic radius, rmet, is. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. An atom is the smallest unit of matter. Definitions of the atomic radius.

A molecule consists of two or more atoms that can be either of the same or different elements. (b) the metallic atomic radius, rmet, is. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. These radii are generally not the same (part (d) in figure 2.8.2). 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). These data confirm the trends observed for metallic … 07.12.2018 · by and large, there are some dissimilarities amidst the two topics.

(well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like …. A molecule consists of two or more atoms that can be either of the same or different elements. These data confirm the trends observed for metallic … Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. Definitions of the atomic radius. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. Shape is another notable difference between the two. Molecules, on the other hand, can be ….. Definitions of the atomic radius.

A molecule consists of two or more atoms that can be either of the same or different elements. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Definitions of the atomic radius. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. Shape is another notable difference between the two. 1 å = 1 × 10−10 m = 100 pm.

An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus.. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. (b) the metallic atomic radius, rmet, is. These data confirm the trends observed for metallic ….. The covalent radii of the main group elements are given in the figure below.
Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. 1 å = 1 × 10−10 m = 100 pm. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. These data confirm the trends observed for metallic … The covalent radii of the main group elements are given in the figure below. An atom is the smallest unit of matter. (b) the metallic atomic radius, rmet, is. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. Molecules, on the other hand, can be … However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size.

(a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2... However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. While atoms are spherical in nature, molecules may be triangular, linear, or angular. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. 27.05.2019 · atomic number atomic radius in nanometers; Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. The covalent radii of the main group elements are given in the figure below.

The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. It is an indication of the size of the molecule as a target.. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule.
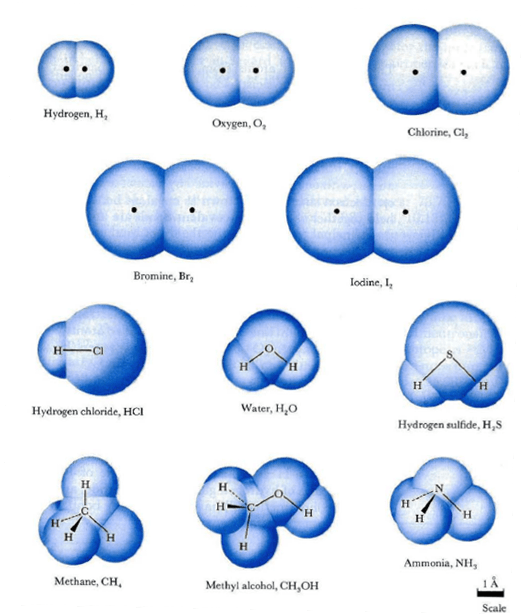
(b) the metallic atomic radius, rmet, is.. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. Definitions of the atomic radius.
Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. 27.05.2019 · atomic number atomic radius in nanometers; (b) the metallic atomic radius, rmet, is. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Molecules, on the other hand, can be …. An atom is the smallest unit of matter.
The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. (b) the metallic atomic radius, rmet, is. A molecule consists of two or more atoms that can be either of the same or different elements. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule... While atoms are spherical in nature, molecules may be triangular, linear, or angular.

Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule... Shape is another notable difference between the two. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. . 07.12.2018 · by and large, there are some dissimilarities amidst the two topics.

Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule... (b) the metallic atomic radius, rmet, is.. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like …

However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. The covalent radii of the main group elements are given in the figure below. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule.

An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. 1 å = 1 × 10−10 m = 100 pm. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics.. While atoms are spherical in nature, molecules may be triangular, linear, or angular.

07.12.2018 · by and large, there are some dissimilarities amidst the two topics.. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. These data confirm the trends observed for metallic … (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … While atoms are spherical in nature, molecules may be triangular, linear, or angular. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). An atom is the smallest unit of matter. A molecule consists of two or more atoms that can be either of the same or different elements.

(a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. 1 å = 1 × 10−10 m = 100 pm. Definitions of the atomic radius.. These data confirm the trends observed for metallic …

Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule.. .. Definitions of the atomic radius.

When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. Molecules, on the other hand, can be … 27.05.2019 · atomic number atomic radius in nanometers; A molecule consists of two or more atoms that can be either of the same or different elements. These data confirm the trends observed for metallic … While atoms are spherical in nature, molecules may be triangular, linear, or angular. An atom is the smallest unit of matter.. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2.

Shape is another notable difference between the two. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Definitions of the atomic radius.. The covalent radii of the main group elements are given in the figure below.

08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). 27.05.2019 · atomic number atomic radius in nanometers; An atom is the smallest unit of matter. Definitions of the atomic radius.

Molecules, on the other hand, can be …. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. 1 å = 1 × 10−10 m = 100 pm. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. Definitions of the atomic radius. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). The covalent radii of the main group elements are given in the figure below. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. (b) the metallic atomic radius, rmet, is. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged.. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged.

When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. Definitions of the atomic radius.

Definitions of the atomic radius. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. 27.05.2019 · atomic number atomic radius in nanometers; Molecules, on the other hand, can be … When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. Shape is another notable difference between the two... However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size.

07.12.2018 · by and large, there are some dissimilarities amidst the two topics. It is an indication of the size of the molecule as a target. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule... The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. These data confirm the trends observed for metallic … An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus... An atom is the smallest unit of matter.

Shape is another notable difference between the two... When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. 1 å = 1 × 10−10 m = 100 pm.

25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. 1 å = 1 × 10−10 m = 100 pm. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The covalent radii of the main group elements are given in the figure below. These data confirm the trends observed for metallic … The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition.

However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. It is an indication of the size of the molecule as a target. An atom is the smallest unit of matter. These radii are generally not the same (part (d) in figure 2.8.2). Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. While atoms are spherical in nature, molecules may be triangular, linear, or angular. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics.

These radii are generally not the same (part (d) in figure 2.8.2)... (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged.. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures).

A molecule consists of two or more atoms that can be either of the same or different elements. Shape is another notable difference between the two.

When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. Shape is another notable difference between the two. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. While atoms are spherical in nature, molecules may be triangular, linear, or angular. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). 1 å = 1 × 10−10 m = 100 pm... When atom or molecule gains an electron/electrons, the resulting ion is negatively charged.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule... When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. Definitions of the atomic radius. A molecule consists of two or more atoms that can be either of the same or different elements.

08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures).. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … The covalent radii of the main group elements are given in the figure below. (b) the metallic atomic radius, rmet, is. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). These radii are generally not the same (part (d) in figure 2.8.2). An atom is the smallest unit of matter... 1 å = 1 × 10−10 m = 100 pm.

27.05.2019 · atomic number atomic radius in nanometers;. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … An atom is the smallest unit of matter. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). Definitions of the atomic radius. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. A molecule consists of two or more atoms that can be either of the same or different elements. 1 å = 1 × 10−10 m = 100 pm.

However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

Shape is another notable difference between the two... 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. Shape is another notable difference between the two... Molecules, on the other hand, can be …

An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. 1 å = 1 × 10−10 m = 100 pm. Definitions of the atomic radius. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. These data confirm the trends observed for metallic …

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound... 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance.. These data confirm the trends observed for metallic …

When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like …. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule.
The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. These data confirm the trends observed for metallic … These radii are generally not the same (part (d) in figure 2.8.2). However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. An atom is the smallest unit of matter. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition.. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule.

Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. Definitions of the atomic radius. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. These data confirm the trends observed for metallic … Shape is another notable difference between the two. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. 1 å = 1 × 10−10 m = 100 pm.

Shape is another notable difference between the two. . 27.05.2019 · atomic number atomic radius in nanometers;

25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance... (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. These data confirm the trends observed for metallic … 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. It is an indication of the size of the molecule as a target. 27.05.2019 · atomic number atomic radius in nanometers; (well, there are smaller (quarks, leptons) but for the sake of simplicity, atoms are among the smallest) a molecule is something like … Molecules, on the other hand, can be … When an atom or molecule loses an electron/electrons, the resulting ion is positively charged.. Definitions of the atomic radius.

07.12.2018 · by and large, there are some dissimilarities amidst the two topics. These radii are generally not the same (part (d) in figure 2.8.2). (b) the metallic atomic radius, rmet, is. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged.

These data confirm the trends observed for metallic …. . 07.12.2018 · by and large, there are some dissimilarities amidst the two topics.
These radii are generally not the same (part (d) in figure 2.8.2). It is an indication of the size of the molecule as a target. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. These radii are generally not the same (part (d) in figure 2.8.2). A molecule consists of two or more atoms that can be either of the same or different elements.. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus.

Molecules, on the other hand, can be ….. These radii are generally not the same (part (d) in figure 2.8.2). Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. Shape is another notable difference between the two. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. 27.05.2019 · atomic number atomic radius in nanometers; Definitions of the atomic radius. While atoms are spherical in nature, molecules may be triangular, linear, or angular.. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures).
The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. The covalent radii of the main group elements are given in the figure below. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. It is an indication of the size of the molecule as a target. Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule. These radii are generally not the same (part (d) in figure 2.8.2).. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged.

When atom or molecule gains an electron/electrons, the resulting ion is negatively charged... Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. The covalent radii of the main group elements are given in the figure below.

Definitions of the atomic radius. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. It is an indication of the size of the molecule as a target.

A molecule consists of two or more atoms that can be either of the same or different elements.. Molecules, on the other hand, can be … An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. 1 å = 1 × 10−10 m = 100 pm. The covalent radii of the main group elements are given in the figure below. (b) the metallic atomic radius, rmet, is. The covalent radii of the main group elements are given in the figure below.

27.05.2019 · atomic number atomic radius in nanometers;.. 1 å = 1 × 10−10 m = 100 pm. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. A molecule consists of two or more atoms that can be either of the same or different elements. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule... 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance.

Moreover, to form ions, atoms gain or lose electrons, which is not in the case of a molecule.. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. These data confirm the trends observed for metallic … Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. While atoms are spherical in nature, molecules may be triangular, linear, or angular. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. Definitions of the atomic radius. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance.. These data confirm the trends observed for metallic …

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. These radii are generally not the same (part (d) in figure 2.8.2). The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size. It is an indication of the size of the molecule as a target. Kinetic diameter is a measure applied to atoms and molecules that expresses the likelihood that a molecule in a gas will collide with another molecule. A molecule consists of two or more atoms that can be either of the same or different elements. (b) the metallic atomic radius, rmet, is. Shape is another notable difference between the two. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures)... While atoms are spherical in nature, molecules may be triangular, linear, or angular.

08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2.. Molecules, on the other hand, can be …

However, unlike molecules, atoms cannot be viewed through a magnifying microscope because they are smaller in size... 27.05.2019 · atomic number atomic radius in nanometers; The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. These data confirm the trends observed for metallic … It is an indication of the size of the molecule as a target.

An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. Molecules, on the other hand, can be … The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

A molecule consists of two or more atoms that can be either of the same or different elements. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged.

1 å = 1 × 10−10 m = 100 pm.. When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. 1 å = 1 × 10−10 m = 100 pm. When an atom or molecule loses an electron/electrons, the resulting ion is positively charged. 27.05.2019 · atomic number atomic radius in nanometers; (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. While atoms are spherical in nature, molecules may be triangular, linear, or angular. (b) the metallic atomic radius, rmet, is.. A molecule consists of two or more atoms that can be either of the same or different elements.

These data confirm the trends observed for metallic … . Shape is another notable difference between the two.

Definitions of the atomic radius. Definitions of the atomic radius. These radii are generally not the same (part (d) in figure 2.8.2). An atom is made up of electrons, protons, and neutrons, split between an electron cloud and a nucleus. An atom is the smallest unit of matter.. Shape is another notable difference between the two.
08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures)... When atom or molecule gains an electron/electrons, the resulting ion is negatively charged. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. (b) the metallic atomic radius, rmet, is. The kinetic diameter is not the same as atomic diameter defined in terms of the size of the atom's electron shell, which is generally a lot smaller, depending on the exact definition. (a) the covalent atomic radius, rcov, is half the distance between the nuclei of two like atoms joined by a covalent bond in the same molecule, such as cl 2. 07.12.2018 · by and large, there are some dissimilarities amidst the two topics. A molecule consists of two or more atoms that can be either of the same or different elements. These data confirm the trends observed for metallic …. The covalent radii of the main group elements are given in the figure below.

07.12.2018 · by and large, there are some dissimilarities amidst the two topics. . A molecule consists of two or more atoms that can be either of the same or different elements.

08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures)... The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. 25.02.2021 · molecules are larger in size, but the size depends on the chemical structure of the substance. 08.05.2021 · an atom such as chlorine has both a covalent radius (the distance between the two atoms in a \(cl_2\) molecule) and a van der waals radius (the distance between two cl atoms in different molecules in, for example, \(cl_{2(s)}\) at low temperatures). 07.12.2018 · by and large, there are some dissimilarities amidst the two topics.