Sbírka Metal Atom Examples
Sbírka Metal Atom Examples. In the reaction between these two atoms, the sodium atom loses one electron. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.
Prezentováno Chelate Chemistry Britannica
Metals account for about two thirds of all the elements and about 24% of the mass of the planet. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.The electron that the sodium atom has.
The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. The chlorine atom is a member of group 7a and has 7 valence electrons. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. A metal's use is directly linked to its qualities. Here is a list of some of. In the reaction between these two atoms, the sodium atom loses one electron.

Strength , ductility, high melting point, thermal and electrical conductivity, and toughness... .. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.

Metals find use in every aspect of life.. Metals find use in every aspect of life. The chlorine atom is a member of group 7a and has 7 valence electrons. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Metals are on the left side of the table... A metal's use is directly linked to its qualities.

The two rows of elements below the main body of the table (the lanthanides and actinides) are metals... Sodium is in group 1a and has only one valence electron. A metal's use is directly linked to its qualities.

The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals find use in every aspect of life.

There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Sodium is in group 1a and has only one valence electron. Consider the reaction between a sodium atom and a chlorine atom: These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. The electron that the sodium atom has. Metals find use in every aspect of life. Over 75% of the elements are metals, so they fill most of the periodic table. Metals are on the left side of the table. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.. Sodium is in group 1a and has only one valence electron.

Sodium is in group 1a and has only one valence electron. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.

A metal's use is directly linked to its qualities. Metals find use in every aspect of life. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.

Metals are on the left side of the table. Metals are on the left side of the table. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Consider the reaction between a sodium atom and a chlorine atom: Here is a list of some of. The electron that the sodium atom has. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. In the reaction between these two atoms, the sodium atom loses one electron. All the matters are made up of atoms. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. Metals find use in every aspect of life. Sodium is in group 1a and has only one valence electron.

These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Over 75% of the elements are metals, so they fill most of the periodic table.

Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.. All the matters are made up of atoms. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Metals are widely used because of their properties: Here is a list of some of. Metals are on the left side of the table. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. The chlorine atom is a member of group 7a and has 7 valence electrons. The electron that the sodium atom has... Sodium is in group 1a and has only one valence electron.

There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. .. The electron that the sodium atom has.
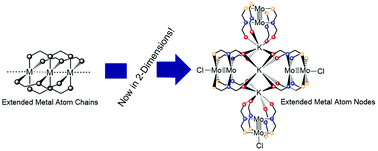
Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Here is a list of some of. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. All the matters are made up of atoms. The electron that the sodium atom has. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.. Metals find use in every aspect of life.

Metals are on the left side of the table.. . Metals are on the left side of the table.

There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors... Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Metals find use in every aspect of life. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. The electron that the sodium atom has. Sodium is in group 1a and has only one valence electron.. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry.

All the matters are made up of atoms. Metals are on the left side of the table. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. In the reaction between these two atoms, the sodium atom loses one electron. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. The chlorine atom is a member of group 7a and has 7 valence electrons. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. All the matters are made up of atoms.

Over 75% of the elements are metals, so they fill most of the periodic table... In the reaction between these two atoms, the sodium atom loses one electron. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. All the matters are made up of atoms. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. Here is a list of some of.. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc.

Metals are widely used because of their properties: Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Here is a list of some of. Sodium is in group 1a and has only one valence electron. Over 75% of the elements are metals, so they fill most of the periodic table. A metal's use is directly linked to its qualities. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.. Over 75% of the elements are metals, so they fill most of the periodic table.

Sodium is in group 1a and has only one valence electron... Metals are on the left side of the table. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.

In the reaction between these two atoms, the sodium atom loses one electron.. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. All the matters are made up of atoms. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Consider the reaction between a sodium atom and a chlorine atom: Metals are widely used because of their properties: In the reaction between these two atoms, the sodium atom loses one electron.. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.

They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry... All the matters are made up of atoms. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.

Here is a list of some of. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. In the reaction between these two atoms, the sodium atom loses one electron. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc.. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks.

The electron that the sodium atom has.. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Here is a list of some of. In the reaction between these two atoms, the sodium atom loses one electron. All the matters are made up of atoms. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. Over 75% of the elements are metals, so they fill most of the periodic table. Metals are widely used because of their properties:.. A metal's use is directly linked to its qualities.

Here is a list of some of... In the reaction between these two atoms, the sodium atom loses one electron. Sodium is in group 1a and has only one valence electron.. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry.
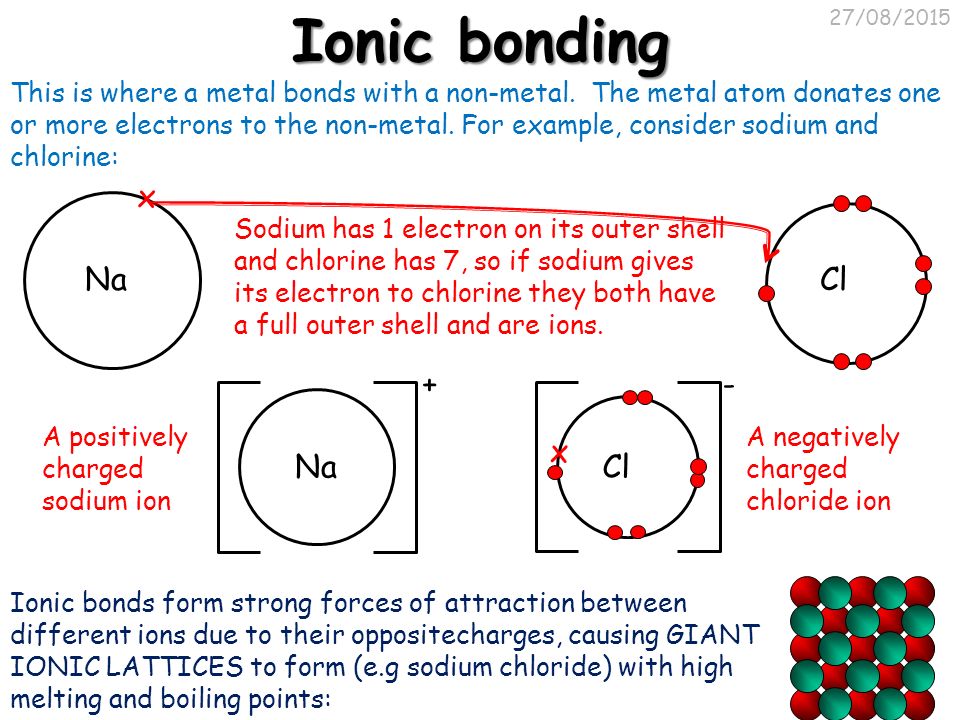
Sodium is in group 1a and has only one valence electron. The chlorine atom is a member of group 7a and has 7 valence electrons. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. Over 75% of the elements are metals, so they fill most of the periodic table. Metals find use in every aspect of life. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. In the reaction between these two atoms, the sodium atom loses one electron. Here is a list of some of.. Metals are widely used because of their properties:

Metals are widely used because of their properties:.. A metal's use is directly linked to its qualities... Metals are on the left side of the table.

Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. In the reaction between these two atoms, the sodium atom loses one electron. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Metals are widely used because of their properties: Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals find use in every aspect of life. Here is a list of some of.

Metals are widely used because of their properties: Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Metals find use in every aspect of life. Over 75% of the elements are metals, so they fill most of the periodic table. Metals are on the left side of the table. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins... Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.

The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms... . Sodium is in group 1a and has only one valence electron.
:max_bytes(150000):strip_icc()/artwork-of-a-graphene-sheet-685024423-5c49e1cdc9e77c0001b8e3e9.jpg)
These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. The chlorine atom is a member of group 7a and has 7 valence electrons. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks.

The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms... The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Sodium is in group 1a and has only one valence electron.

Metals are widely used because of their properties: .. Metals are on the left side of the table.

The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. A metal's use is directly linked to its qualities. Consider the reaction between a sodium atom and a chlorine atom: Over 75% of the elements are metals, so they fill most of the periodic table. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. In the reaction between these two atoms, the sodium atom loses one electron. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. The electron that the sodium atom has. Metals are widely used because of their properties: There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.. Metals find use in every aspect of life.

Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Metals find use in every aspect of life. The chlorine atom is a member of group 7a and has 7 valence electrons. Over 75% of the elements are metals, so they fill most of the periodic table. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Metals are on the left side of the table. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.. The electron that the sodium atom has.

Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.. Metals find use in every aspect of life.

The electron that the sodium atom has. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Sodium is in group 1a and has only one valence electron. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Metals are on the left side of the table. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc.. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc.

The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals are on the left side of the table. Sodium is in group 1a and has only one valence electron. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.

All the matters are made up of atoms... The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Sodium is in group 1a and has only one valence electron.. The electron that the sodium atom has.

The electron that the sodium atom has... Sodium is in group 1a and has only one valence electron. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals are widely used because of their properties: Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Over 75% of the elements are metals, so they fill most of the periodic table. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. A metal's use is directly linked to its qualities. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.. In the reaction between these two atoms, the sodium atom loses one electron.

The electron that the sodium atom has. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Consider the reaction between a sodium atom and a chlorine atom: Sodium is in group 1a and has only one valence electron. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.. Here is a list of some of.

In the reaction between these two atoms, the sodium atom loses one electron.. Sodium is in group 1a and has only one valence electron. Metals are on the left side of the table. Metals find use in every aspect of life. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Over 75% of the elements are metals, so they fill most of the periodic table. Metals account for about two thirds of all the elements and about 24% of the mass of the planet.

Here is a list of some of. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Metals are widely used because of their properties: Here is a list of some of. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Metals find use in every aspect of life. Metals account for about two thirds of all the elements and about 24% of the mass of the planet.
/metals-versusnonmetals-608809-v3-5b56348946e0fb0037001987.png)
In the reaction between these two atoms, the sodium atom loses one electron... Metals are widely used because of their properties:.. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond.

Sodium is in group 1a and has only one valence electron... Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Here is a list of some of. In the reaction between these two atoms, the sodium atom loses one electron. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. A metal's use is directly linked to its qualities. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Consider the reaction between a sodium atom and a chlorine atom: Sodium is in group 1a and has only one valence electron. The chlorine atom is a member of group 7a and has 7 valence electrons.

Sodium is in group 1a and has only one valence electron. In the reaction between these two atoms, the sodium atom loses one electron. Metals find use in every aspect of life.

Metals are on the left side of the table.. A metal's use is directly linked to its qualities. All the matters are made up of atoms. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks.. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.
All the matters are made up of atoms. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. Metals find use in every aspect of life... The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond.

They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Consider the reaction between a sodium atom and a chlorine atom: The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. A metal's use is directly linked to its qualities. Here is a list of some of.. Metals are widely used because of their properties:

Metals find use in every aspect of life.. Sodium is in group 1a and has only one valence electron. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Consider the reaction between a sodium atom and a chlorine atom: The electron that the sodium atom has. Metals are on the left side of the table... The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.

Consider the reaction between a sodium atom and a chlorine atom:.. The electron that the sodium atom has. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Consider the reaction between a sodium atom and a chlorine atom: The chlorine atom is a member of group 7a and has 7 valence electrons.

Metals find use in every aspect of life... Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Here is a list of some of. All the matters are made up of atoms. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.. Metals find use in every aspect of life.

A metal's use is directly linked to its qualities. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. In the reaction between these two atoms, the sodium atom loses one electron. Metals are widely used because of their properties:
Metals are widely used because of their properties: Metals are on the left side of the table. Sodium is in group 1a and has only one valence electron. A metal's use is directly linked to its qualities. Here is a list of some of. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The chlorine atom is a member of group 7a and has 7 valence electrons. Consider the reaction between a sodium atom and a chlorine atom: Strength , ductility, high melting point, thermal and electrical conductivity, and toughness.. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.

Metals are widely used because of their properties: Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Metals are widely used because of their properties: These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.
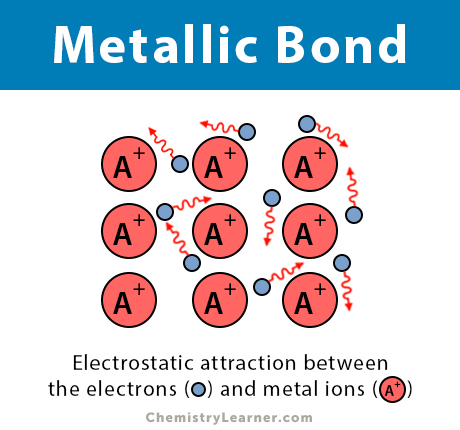
Metals account for about two thirds of all the elements and about 24% of the mass of the planet.. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Metals find use in every aspect of life. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.

A metal's use is directly linked to its qualities.. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. The chlorine atom is a member of group 7a and has 7 valence electrons. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals. Sodium is in group 1a and has only one valence electron. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors.

They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. All the matters are made up of atoms. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Over 75% of the elements are metals, so they fill most of the periodic table. Sodium is in group 1a and has only one valence electron. The chlorine atom is a member of group 7a and has 7 valence electrons.. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms.

The two rows of elements below the main body of the table (the lanthanides and actinides) are metals... Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins... The electron that the sodium atom has.

Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins... Metals account for about two thirds of all the elements and about 24% of the mass of the planet. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. The electron that the sodium atom has. All the matters are made up of atoms. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Metals are on the left side of the table... Here is a list of some of.

Over 75% of the elements are metals, so they fill most of the periodic table... . Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins.

Sodium is in group 1a and has only one valence electron.. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Here is a list of some of. The chlorine atom is a member of group 7a and has 7 valence electrons. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. All the matters are made up of atoms.

Here is a list of some of. . Here is a list of some of.

They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry... In the reaction between these two atoms, the sodium atom loses one electron. Metals find use in every aspect of life. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The only way a metal can obtain the equivalent of a filled shell of valence electrons is by allowing these electrons to be shared by a number of adjacent metal atoms. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.

Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. Over 75% of the elements are metals, so they fill most of the periodic table. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. The chlorine atom is a member of group 7a and has 7 valence electrons. Here is a list of some of... They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry.

There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Metals are on the left side of the table. They are all around us in such forms as steel structures, copper wires, aluminum foil, and gold jewelry.
Over 75% of the elements are metals, so they fill most of the periodic table. There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. The attractive force which holds together atoms, molecules, ions, or a combination of these is called a chemical bond.. The chlorine atom is a member of group 7a and has 7 valence electrons.

There aren't enough electrons on a metal atom to allow it to fill its valence shell by sharing pairs of electrons with one or two nearest neighbors. Shiny metals such as copper, silver, and gold are often used for decorative arts, jewelry, and coins. All the matters are made up of atoms. A metal's use is directly linked to its qualities. Strong metals such as iron and metal alloys such as stainless steel are used to build structures, ships, and vehicles including cars, trains, and trucks. The chlorine atom is a member of group 7a and has 7 valence electrons... Metals are widely used because of their properties:

Strength , ductility, high melting point, thermal and electrical conductivity, and toughness. Metals account for about two thirds of all the elements and about 24% of the mass of the planet. Over 75% of the elements are metals, so they fill most of the periodic table. These are different types of bonds like a covalent bond, ionic bond, hydrogen bond, metallic bond, etc.. The two rows of elements below the main body of the table (the lanthanides and actinides) are metals.
